Acute myeloid leukemia (AML) is a heterogeneous disease associated with poor survival and frequent relapse rates due to the inability to effectively target leukemic stem cells (LSCs) and acquired resistance to existing treatments. Development of treatments targeting LSCs and overcoming acquired resistance is crucial to improve the survival of AML patients. The PI3 kinase (PI3K)/AKT signaling pathway is activated in 80% of AML patients, across various AML subtypes, making PI3K an attractive therapeutic target for this disease. Several PI3K inhibitors, such as copanlisib, are clinically approved for other indications, with a reasonable toxicity profile. The effects of PI3K inhibitors on LSCs however, remains unknown.
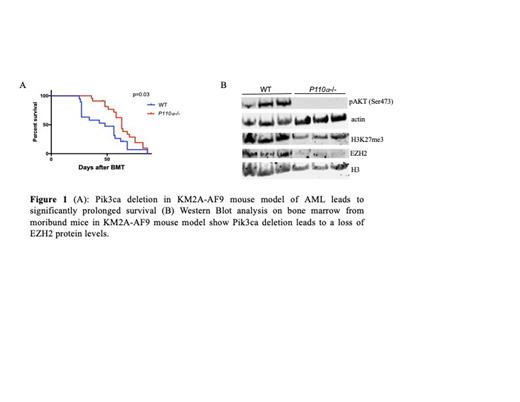
Several catalytic isoforms of PI3K are expressed in hematopoietic cells. To determine whether these PI3K isoforms play a role in LSC self-renewal, we performed colony assays on KMT2A-AF9 retrovirally transduced hematopoietic stem and progenitor cells (HSPCs) with either genetic deletion or pharmacologic inhibition of PI3K isoforms. We found that disruption of PI3K signaling reduces the self-renewal of LSCs and promotes myeloid differentiation in both mouse LSCs and human AML cell lines. KMT2A-AF9 induces leukemic transformation via aberrant upregulation of key self-renewal genes in hematopoietic progenitors, such as HoxA9 and Meis1 (PMID: 16862118). We found that pharmacologic inhibition of PI3K isoforms leads to a reduction in HoxA9 and Meis1 expression in LSC colonies. In the KMT2A-AF9 retroviral bone marrow transplantation model of AML, genetic deletion of the PI3K isoform Pik3ca leads to a reduction in LSC numbers in the bone marrow and prolonged survival (Fig 1A), although it is not curative. In addition, we observed significant prolongation of survival upon secondary transplantation of Pik3ca leukemic cells, suggesting a reduction in functional LSCs. Transcriptome analysis on LSCs isolated from pre-clinical bone marrow aspirates of KMT2A-AF9 primary transplant mice revealed downregulation of the target genes of HoxA9, an important driver of LSC self-renewal. We also observed enrichment of the targets of EZH2, a histone methyltransferase that catalyzes the H3K27 trimethylation repressive mark. AKT is known to phosphorylate EZH2 at Ser21, leading to inhibition of EZH2 function (PMID: 16224021). Therefore, we hypothesized that Pik3ca deletion can promote LSC differentiation by de-repressing EZH2 activation via AKT inactivation.
To determine whether PI3K inactivation differentiates KMT2A-AF9 leukemic cells via dephosphorylation of EZH2, we performed Western blot analysis on bone marrow cells from Pik3ca KO and WT leukemic mice. As expected, Pik3ca deletion significantly reduced AKT phosphorylation. Unexpectedly, a reduction of total EZH2 protein levels was also observed in Pik3ca KO leukemic cells (Fig 1B), suggesting that EZH2 downregulation may be a non-genetic acquired resistance mechanism to PI3K inactivation. Histone proteomics analysis at two different timepoints after treatment with the PI3K inhibitor copanlisib revealed rapid and dynamic changes in histone modifications, including H3K27me3 and H3K4me3, consistent with epigenetic mechanisms of resistance to PI3K inhibition. EZH1, a homolog of EZH2, has been reported to compensate for EZH2 function (PMID: 26219303, PMID: 31189531). Thus, we hypothesize that PI3K inhibition may sensitize leukemic cells to EZH1/2 dual inhibition by reducing EZH2 levels. We found that dual inhibition with copanlisib and the EZH1/2 inhibitor valemetostat leads to a combinatorial effect to strongly decrease the proliferation of AML cell lines and AML patient samples. Additionally, we found that treatment of KMT2A-AF9 AML mice with these compounds induces myeloid differentiation and leads to a reduction in leukemia initiating cell capacity, as demonstrated by prolonged survival upon secondary transplantation. Overall, our data suggests that PI3K plays an important role in leukemic stem cells, and that EZH2 downregulation is a non-genetic mechanism of resistance to PI3K inhibition. Additionally, our findings indicate that combining PI3K inhibitors with EZH1/2 dual inhibitors could be a promising therapeutic approach to reduce LSCs, prevent resistance, and prolong survival in AML patients.
Disclosures
Verma:BMS: Research Funding; GSK: Research Funding; Eli Lilly: Research Funding; Stelexis: Current equity holder in private company, Honoraria, Other: Scientific Advisor; Bakx: Current equity holder in private company, Other: Scientific Advisor; Novartis: Other: Scientific Advisor; Acceleron: Other: Scientific Advisor; Celgene: Other: Scientific Advisor; Curis: Research Funding; Medpacto: Research Funding; Incyte: Research Funding; Prelude: Research Funding; Janssen: Honoraria; Throws Exception: Current equity holder in private company. Gritsman:ADC Therapeutics: Research Funding; iOnctura: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal